FREQUENTLY ASKED QUESTIONS
FRIZZLE (FRRS1L) INFORMATION
ORGANIZATION
Organization name: Finding Hope for FRRS1L
Website: www.FRRS1L.org
Contact: contact@FRRS1L.org
EIN: 87-3285525
Mailing Address:
Finding Hope for FRRS1L
301 BOARDWALK DR, PO Box 270405
FORT COLLINS, CO 80525-9998
What is Finding Hope for Frizzle (FRRS1L)?
Finding Hope for Frizzle (FRRS1L) is a registered 501(c)(3) nonprofit organization based in Colorado. It was founded in 2021 by parents of children affected by FRRS1L (Frizzle) genetic disease who came together with a shared mission: to develop a life-changing treatment.
The organization is entirely volunteer-run, with a board of six directors and additional officer roles — none of whom are paid. Every dollar raised goes directly toward developing gene replacement therapy for Frizzle (FRRS1L).
Finding Hope for Frizzle holds the rights to its treatment program and is fully funding and leading its development. The organization plans to serve as the sponsor of an Investigational New Drug (IND) application with the U.S. Food and Drug Administration (FDA), with the goal of beginning clinical trials for patients before the end of 2026.
What is the mission of Finding Hope for Frizzle (FRRS1L)?
Finding Hope for Frizzle (FRRS1L) has three mission areas:
Frizzle Community: To create a community of Hope for Frizzle (FRRS1L) genetic disease children and families to connect, communicate, collaborate, and support one another.
Treatment: To fund the research and development of gene replacement therapy — an approach that has restored function in FRRS1L mouse models and offers real hope for future treatment.
Awareness: To share information and resources with the public, medical professionals, and researchers to increase and understanding and awareness of Frizzle (FRRS1L) disease.
FRIZZLE (FRRS1L) DISEASE
FRRS1L
FRRS1L stands for: Ferric Chelate Reductase 1 Like.
How do you pronounce the name of the disease?
The disease is pronounced “Frizzle”.
Is it a Developmental and Epileptic Encephalopathy (DEE)?
Yes, Frizzle is a Developmental and Epileptic Encephalopathy (DEE) type 37.
Is it a neurological disease?
Yes, Frizzle is a neurological disease.
Is Frizzle genetic disease autosomal recessive?
Yes. Frizzle genetic disease is an autosomal recessive disease. It requires two inherited non-functioning FRRS1L genes to be present in a patient to have the disease.
Do carriers have symptoms?
No. Carriers of a nonfunctioning FRRS1L gene do not have symptoms. It takes two nonfunctioning FRRS1L genes to have disease progression and symptoms.
What is Frizzle (FRRS1L) genetic disease?
FRRS1L disease (pronounced “Frizzle”) is a rare genetic disease caused by a loss of function mutation in the FRRS1L gene. This gene is responsible for encoding a protein that plays a critical role in ensuring normal development and function of the brain. The loss of function of the FRRS1L gene interferes with proper building of the AMPA receptors in the brain, which are essential for neurons in the brain to communicate with each other. In individuals with Frizzle disease, disruptions in these neural signaling pathways result in severe epilepsy, movement disorder, regression, loss of function, additional health issues due to loss of function, and lifelong disability.
In simple terms: To have Frizzle (FRRS1l) disease a patient has two bad copies of the FRRS1L gene and they do not work or function. This causes the messages in the brain to not be successfully sent, resulting in seizures, regression, and loss of function. The child loses all motor function. Children are trapped in a body they can’t move but they are still inside and have cognitive understanding.
Frizzle Disease progression:
Mothers of Frizzle (FRRS1L) patients report a normal pregnancy. Most children with Frizzle disease re born appearing healthy.
Around 6 months of age, developmental delays begin to appear.
Between 1 and 2 years old, children often experience sudden and severe regression, along with the onset of seizures.
Over time, the condition progresses to include:
Severe epileptic encephalopathy (uncontrolled epilepsy)
Movement disorders
Profound developmental delays
Low muscle tone (hypotonia)
Cerebral palsy
Feeding and swallowing difficulties; difficulty with airway clearance
Respiratory complications and failure
Loss of motor control: unable to chew or swallow; they can’t hold their head up, sit, stand or walk; they can’t hold things with their hands; they can’t lift their arms; they can’t turn their head easily from side to side.
Complete physical dependence on parents/guardians/caregivers for all movement, care, and nutrition.
They require support from medical devices such as positioning equipment, feeding tubes, suctioning, respiratory clearance, orthotics, bracing, and ventilation.
Cognitive Awareness and Capability:
Despite the loss of function and control, FRRS1L disorder children do maintain cognitive awareness, showing their ability to understand and learn. They still present clear emotional responses and communication through sounds and facial expressions. They convey pain, boredom, frustration, happiness, and joy. They can communicate using different eye-gaze speech methods – they can look at options or hand signals to make decisions, or some children even use an eye-gaze communication device where they can communicate effectively with their eyes. Eye gaze communication has shown in those patients cognitive understanding, awareness, and learning.
Seizures/Epilepsy
Current epileptic medication for Frizzle disease is ineffective and limited to symptom reduction and not management or control. Children’s seizures are resistant to epileptic medicines, which means they suffer from ongoing status epilepticus.
Is Frizzle genetic disease life limiting and life threatening?
Yes. Due to the rapid regression and extensive loss of function, Frizzle patients’ lives are at risk. Threat to life can include but not be limited to inability to eat/drink/nutrition, airway blockage, respiratory failure, pneumonia, and epilepsy.
What does care for Frizzle (FRRS1L) genetic disease require?
Due to loss of function and abilities, Frizzle (FRRS1L) genetic disease patients require 24/7 care intensive skilled nursing care, support, and monitoring. They require support from medical devices such as tube feedings, suctioning, respiratory clearance, chest compressions, cough assist, oxygen, and ventilation; as well as special equipment for body positioning.
Are there any medications to stop regression or reverse regression; stop/manage epilepsy?
No. There are no effective medications available for FRRS1L disorder. So far, no current drugs can prevent regression, improve development or successfully manage seizures in these patients. Patients are highly refractory to seizure medications.
When was Frizzle (FRRS1L) discovered as a genetic disease?
Frizzle (FRRS1L) genetic disorder was discovered in 2016 research.
How are patients identified and/or diagnosed with Frizzle (FRRS1L)?
Diagnosis of Frizzle (FRRS1L) disease is achieved through genetic testing. To date, basic genetic testing doesn’t include the FRRS1L gene, so whole exome or whole genome sequencing is required for diagnosis. Increased awareness of Frizzle around the world could help doctors include the FRRS1L gene in specific genetic panels in the future.
What is prevalence of Frizzle?
Currently there are 100+ known cases identified; however, this number is growing monthly as patients pursue whole genome sequencing testing and because of the increase in awareness of the disease. Frizzle (FRRS1L) genetic disease is found across multiple people groups all around the world. Due to its recent discovery in 2016, and because of limited access to genetic testing, Frizzle (FRRS1L) disease is highly likely to be underdiagnosed and/or misdiagnosed in patients. Most new cases are being found in babies due to early intervention and genetic testing.
Is Frizzle a rare disease?
Yes. Frizzle (FRRS1L) disease is currently considered a rare disease.
Where is Frizzle (FRRS1L) disease found around the world?
Frizzle (FRRS1L) disease has been found around the world, and in multiple people groups. Currently there are patients in: Afghanistan, Bulgaria, Croatia, Germany, India, Iran, Italy, Middle East, Netherlands, Puerto Rico, South Korea, Turkey, and the USA.
TREATMENT DEVELOPMENT INFORMATION
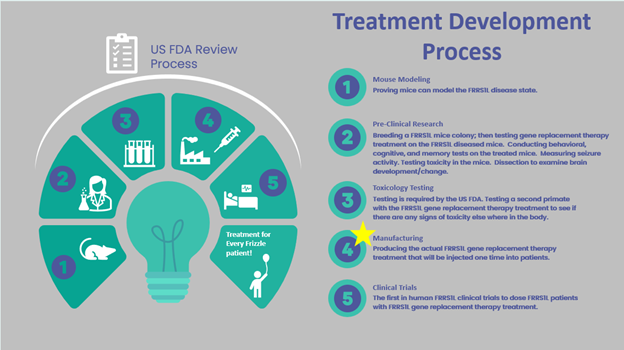
What is the treatment currently being developed for Frizzle (FRRS1L) disease? How does it work?
Finding Hope for Frizzle is developing gene replacement therapy using the TfR1 CapX technology. The TfR1 CapX capsid will contain the FRRS1L transgene. The treatment will be administered intravenously (IV) to a patient. The TfR1 CapX capsid will cross the Blood Brain Barrier (BBB) into the cells of the brain providing broad distribution to the brain. The treatment is gene therapy and thus can only be given as a onetime dose to patients. The hope of the organization is that once the good copies of Frizzle gene are delivered to the brain cells, the result will be the rebuilding of functioning AMPA receptors, and the return of function to the brain and body of patients.
Why is it a one-time dose for a Frizzle (FRRS1L) diseased patient?
Gene therapies use a virus capsid as the mechanism to carry the good copy of the Frizzle gene into the brain. All viruses cause an immune response in the body. Even though our treatment is beneficial, with no active virus DNA inside, the patient’s body still views the virus capsid as a threat to the body and begin to attack the virus. This creates an immune response in the patients and ultimately creates antibodies to protect against the virus in future encounters for the body. Thus, gene therapy cannot be re-dosed because antibodies created for a first dose would prevent any treatment from being affective again in the body. The antibodies would stop the treatment.
How will Frizzle (FRRS1L) disease treatment be administered?
Frizzle (FRRS1L) disease treatment will first be provided to patients in a clinical trial. The treatment will be administered intravenously (IV).
When will treatment be available?
The organization is currently in the manufacturing stage of developing the treatment. The organization is 100% responsible for paying for every aspect of the treatment development process. If the remaining funds needed ($4 million) can be raised, then the goal is to begin a clinical trial by September 2026.
Where is the organization in the process for treatment development?
The organization is currently in the manufacturing stage of developing the treatment. The organization is the sponsor of its IND with the US FDA and are in current communications with the US FDA.
When will a clinical trial begin?
The organization has a goal to begin a Phase I/II clinical trial by September 2026; however, this is subject to change based on raising the funds needed to pay for the trial. Since this is a novel form of therapy, patients will be recruited at intervals to ensure no serious drug related adverse events appear.
Where will the clinical trial be held?
The organization is waiting for FDA confirmation before providing additional information on a clinical trial. Please check back for future updates.
Who will be eligible for the clinical trial?
Following FDA guidance, the clinical site will choose patients based on a strict set of eligibility criteria that ensure safety and maximize the ability to assess the efficacy of gene therapy.
Why did the organization choose to use the novel TfR1 CapX technology for its treatment?
As a parent-led organization that has control of our treatment program, we made the decision to develop our gene replacement therapy for Frizzle genetic disease using the TfR1CapX technology. The research from Dr. Ben Deverman and his lab on TfR1 CapX is incredible. We know our kids and patients can only get dosed once with gene therapy, and we need it to be the best technology possible that can deliver our Frizzle gene to the whole brain. We also need it to be a route of administration that is less invasive and more easily accessible for our global patient population. CapX does exactly that: it is dosed intravenously and can deliver genes throughout the whole brain. Please watch the Frizzle TfR1 CapX webinar for more information.
Who created TfR1 CapX?
Dr. Ben Deverman at the Broad Institute, along with his lab developed the TfR1 CapX capsid technology.
What is TfR1 CapX?
Please see Apertura Gene Therapy to learn more about TfR1 CapX.
RESEARCH INFORMATION
What research has been conducted for Frizzle (FRRS1L) disease treatment?
Gene replacement therapy for Frizzle is currently in development. Finding Hope for Frizzle funded pre-clinical research to test gene replacement therapy on FRRS1L bred and diseased mice. The Frizzle study proved that FRRS1L-bred and diseased mice that received gene replacement treatment successfully regained function, had the rebuilding of AMPA receptors in the mice brains, and had greatly reduced seizure activity. Scientists and researchers believe Frizzle gene replacement therapy has the potential to be life changing for Frizzle (FRRS1L) disease patients, and hopefully restorative for function.
Where can I find more published research on Frizzle (FRRS1L) disease?
A full list of published research is located on our website under the research option.
FUNDRAISING
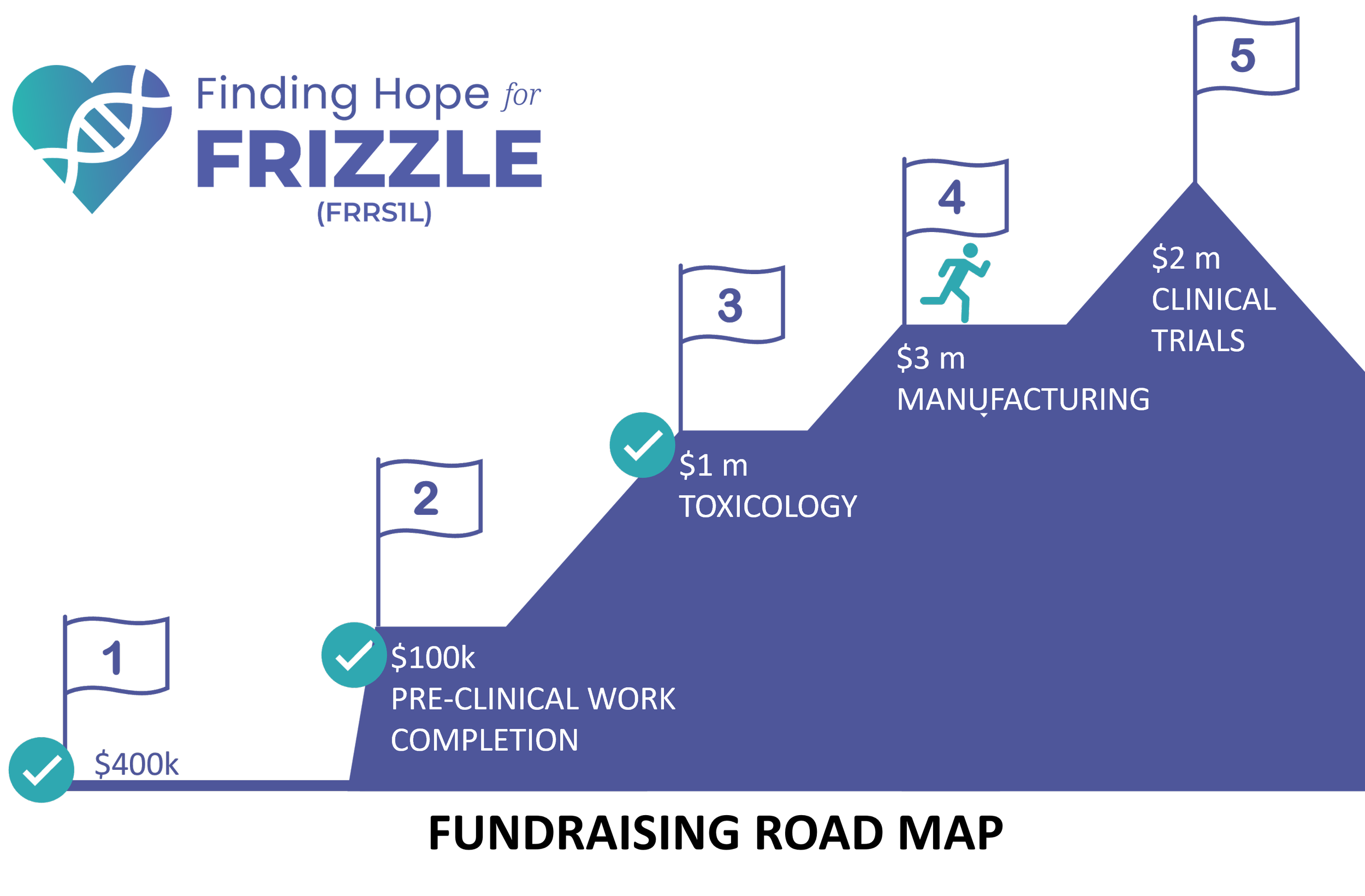
What are the fundraising goals for Finding Hope for Frizzle (FRRS1L)?
Finding Hope for Frizzle has raised $2.5 million to date for the treatment development process. The organization is a nonprofit that raises funds to directly pay for each set of the treatment development process. The remaining need is $4 million, which is needed is to fund the rest of the manufacturing of the actual drug to be given to patients, as well as to fund a clinical trial. Please see the fundraising plan for more details. See graphic 2.
Graphic 2.